愛車的人,換配件時往往想選原廠配件;常吃藥的人,往往也希望吃原廠的藥。人們對「仿製品」的不信任感自然而然。原廠藥 和學名藥 的藥效爭議 ,幾乎從來沒有斷過。
吃藥的人大多知道,很多藥都有兩個「版本」:原廠藥 和學名藥 。
一種藥的原廠藥,是由原始開發這個藥的藥廠生產的。這個藥研發出來、由美國食品藥物管理局(FDA)批准使用後,一般有20年的專利保護期。在這期間,其它藥廠都不可以生產該藥物。當專利期一過,限制一除,其它藥廠就開始紛紛生產這種藥,這就是學名藥。
這樣聽起來,學名藥就是原廠藥的「仿製品」,是其它藥廠照著專利品「仿製」的;而且,學名藥的價錢還比原廠藥便宜80~85%,所謂「便宜沒好貨」,更讓人覺得難以放心使用。也有很多病人表示,從原廠藥換成學名藥後,感覺療效變差、或出現了副作用。
學名藥和原廠藥到底有什麼差別?民眾可以安心服用學名藥嗎?本文中,「健康1+1」帶大家探究這兩種藥的真面目。
學名藥和原廠藥,藥效一樣嗎? 在原廠藥的專利過期後,其它藥廠就可以開始生產學名藥。但不是哪家藥廠都能隨便生產、上市的,必須通過FDA的審核批准。
FDA對於學名藥,有著嚴苛的要求。
首先,學名藥的製造廠的條件,必須滿足FDA的要求。工廠的生產環境、生產設備、藥物包裝等,需和原廠藥的製造廠滿足同等標準。事實上,很多學名藥的製造廠和原廠藥是同一個。
藥物本身更是如此。學名藥在批准生產上市前,必須通過FDA的審核。FDA規定,學名藥和原廠藥必須有同樣的有效成分、劑量、藥性強度、服用方式和血中濃度。
而這個「血中濃度」,是決定藥效的關鍵。
人吃了藥以後,藥在血液裡需要達到一定的有效治療濃度,才能產生效果。瑪摩利醫院癌症 中心腫瘤內科主治醫生黃夷伍說,一般FDA要求兩種藥在血中濃度的差別不能超過10%,這樣在療效上就不會有太大差別。
據FDA調查,原廠藥與學名藥的血中濃度平均差別為3.5%,遠遠低於10%。而這種差別在不同品牌的同種藥物之間也存在,是正常現象,不會引起藥效上的不同。即便這3.5%會引起細微差別,對每個病人來說是原廠藥還是學名藥效果好,也未可知。
安康寧藥房藥劑師洪瑤純表示,有些時候,當專利期過後,其它藥廠還在準備學名藥審核時,原始藥廠會搶先生產自己的學名藥、搶占市場。 「這樣對病人其實很好。」洪瑤純說,這種學名藥和它的原廠藥名字雖然不同,但外觀、成分是一模一樣的。藥劑師通常為換藥的病人選擇這種,讓病人服藥時心裡感覺更踏實。
外觀不是重點 學名藥和原廠藥外觀不同、名字不同,顏色、味道也可能不同。(Shutterstock) 學名藥和原廠藥外觀不同、名字不同,顏色、味道也可能不同,這讓很多民眾以為自己用了「冒牌貨」。
美國商標法不允許學名藥的外觀、名字和原廠藥一模一樣。畢竟這些都是原廠藥的專利,如果都複製去了,倒真成了山寨版。而其它藥廠也要宣傳自己的品牌,自然不會去照搬原廠藥的模式。
藥物的顏色、形狀、口味等,都屬於藥物的非活性成分,這些因素通常不會影響藥物的藥效。
價錢差萬里 學名藥的價錢往往比原廠藥低80~85%。特別對於「天價」藥來說,學名藥減輕了人們花費的負擔,卻也引發了一分錢一分貨的質疑。
洪瑤純舉例,一種降血壓的原廠藥Tenormin,30粒要432美元,但其學名藥Atenolol,30粒只要十幾美元。
為什麼學名藥比原廠藥便宜這麼多?因為原藥廠在開發藥物的時候,需要做各種研究、動物試驗、臨床試驗,從研發到上市平均歷時12年,耗時、耗資巨大。在藥物上市後,還要做市場營銷。而這些花掉的錢,就在賣藥的時候賺回來。
相比之下,學名藥就沒有這麼大的投資壓力。因此,售價也低很多。加之不同品牌學名藥之間的市場競爭,也造成價格不斷降低。
黃夷伍說,在美國,80%的藥品都是學名藥。因為使用學名藥,美國人每星期省了30億美元,一年省去1560億美元。
對學名藥的 4 個質疑,是否有根據 一、病人換藥後,出現種種問題? 一些病人在從原廠藥換成學名藥後,感覺療效變差,或出現一些原來沒有的副作用。(Shutterstock) 既然原廠藥和學名藥沒有療效上的差別,為什麼一些病人在從原廠藥換成學名藥後,感覺療效變差,或出現一些原來沒有的副作用?
發生類似情況,可能由於以下幾種原因:
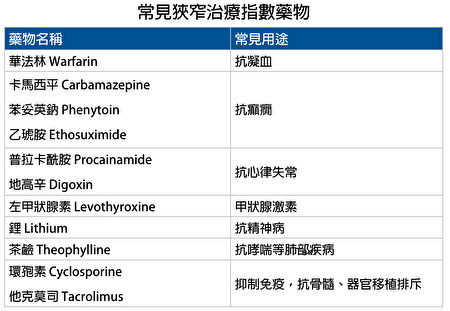
● 狹窄治療指數藥物
上文提到,學名藥和原廠藥之間的血中濃度差異只有3.5%,不會影響療效。但對於一些特殊的藥物,這極小的差異,就可能對病人達不到有效的治療濃度、或造成毒性副作用。這類特殊藥物被稱為「狹窄治療指數藥物」。
黃夷伍表示,這種現象並不侷限於從原廠藥換到學名藥,只要是從一種品牌換到另一種品牌,就可能出現問題。
洪瑤純說,她給病人開甲狀腺素時,就非常謹慎。「如果顧客第一次來,配的是甲狀腺素的藥,我會問,你以前有沒有吃過這種藥,吃的是什麼牌子?因為我們都要給一樣的牌子。」
如果病人吃的是原廠藥,她就會給病人開原廠藥。如果保險不包原廠藥費用,病人自己又負擔不起,那麼她會給病人一次性換成學名藥,但以後就固定使用同一種學名藥,不再更換。
以下是常見的狹窄治療指數的藥物。黃夷伍強調,對於這類藥品,如果換了品牌,要及時告訴醫生,觀察副作用和療效是否有變化:
常見狹窄治療指數藥物。(大紀元製圖) ● 心理作用和身體適應力
病人的心理作用也是造成療效變化的主要原因之一。本來,從一種藥換到另一種藥,病人心理就需要做一些調適。而當保險不再付原廠藥,病人不得不使用學名藥時,心理上的落差也可能加重對學名藥的不信任,服藥後就會感覺身體「不對勁」。同時,一種藥用久了,身體可能已適應了它,這時再換藥,細微的改變也可能使身體出現一些反應。
洪瑤純說,有一個病人,她之前一直給他開其它藥廠生產的學名藥。一次,原始藥廠生產的學名藥有貨了,於是她給這位病人換成了原藥廠的學名藥。
病人過兩天回來對她說:「唉,上次那個藥廠比較好,這個藥廠比較不好。」
洪瑤純笑著回憶:「我跟他說,『可是這次這個藥廠是原廠欸,你真的確定比較不好嗎?』他說,『哦是嘛?那好吧,那我試試看。』」
洪瑤純說,這可能是病人的心理因素,也有可能是病人的身體已經習慣了原來那個藥廠的藥。
美國西奈山醫院Ruttenberg治療中心醫療主任曹哲凱說,有的病人吃原廠藥感覺很好,換了學名藥覺得效果不夠好;還有病人吃慣了學名藥,換了正廠藥也覺得不適應。兩方面都有。「其實醫生很難做評判,我會請病人做評估。」
● 病人自身的情況
有的病人不止吃一種藥,而是同時服用多種藥物。多重藥物在體內作用,就使體內環境變得複雜。因此,當從一種牌子的藥換成另一種——無論是學名藥還是原廠藥,藥物的非活性成分發生細微的變化時,也可能對個別病人造成影響。
曹哲凱建議,如果病人在吃很多種不同的藥,今天換了一個藥,需要請醫生幫你查這些藥有沒有相衝到、會不會影響其它藥的藥效。如果吃了藥身體不適,應儘快給醫生打電話詢問。
無論是哪種原因,出了問題應告知醫生或藥劑師,請他們找出原因,或幫忙調整藥物。
VIDEO
二、學名藥的「非活性成分」與原廠藥不同,會對療效造成影響 除了改變藥物的外觀、味道外,非活性成分的作用主要是幫助藥物的吸收,也就是確保藥物中的有效成分發揮效用。而當FDA審核藥物時,看的是最終藥物在血液中的濃度、最終療效。
比如,有些非活性成分幫助藥物逃過胃酸侵蝕,順利進入小腸後再釋放出來。「所以不管藥是怎麼配的,服進去了在血裡達到了同樣的治療濃度,它的療效就是一樣的。」黃夷伍說。
三、很多學名藥外包給印度等地生產,生產質量難以保障 FDA規定學名藥工廠的生產環境、生產設備、藥物包裝等,需和原廠藥的製造廠滿足同等標準。(Shutterstock) 為降低成本,很多學名藥將藥物外包給印度等地的工廠生產。黃夷伍表示,只要藥物在美國上市,生產環境和藥物質量就必須經由FDA審批,病人不用擔心,「原廠藥的很多主要成分也是在其它國家製造的。」
雖然如此,藥廠造假的現象依然是存在的。2013年,印度藥廠Ranbaxy被陸續查出學名藥臨床數據造假、藥品中發現頭髮、員工上完洗手間後不洗手等問題。隨後,Ranbaxy的莫哈里藥廠被禁止生產FDA管控的藥物,直到藥廠符合FDA要求。
四、 FDA 審查過後,如何確保後續生產的質量不變? 在學名藥被批准後,FDA還會定期檢查該藥的生產環境和藥品質量。另外,FDA有嚴格的副作用和療效報告系統。藥品上市後,如果出現較大的副作用,或是病人、醫生反饋有問題,FDA會調查該藥品,並作出相應修整措施。
保險不付,怎麼辦? 很多病人使用學名藥,也是迫於原廠藥的費用壓力。當一種原廠藥有了相應的學名藥,保險公司就會停止給付原廠藥的費用。病人如果要使用原廠藥,就必須自己支付差價。洪瑤純指出,如果病人使用學名藥出現不良效果,醫生可以幫病人向保險公司申請,要求保付原廠藥的費用。
如果一種原廠藥很貴,即便尚未有學名藥出現,有些保險公司也可能讓病人先使用價格較低類似藥物。但如果病人必須使用原廠藥,用其它藥物都會產生不良效果,也可以請醫生幫忙向保險公司申請。
學名藥怎麼找? 如果病人覺得原廠藥價格負擔重,希望使用價格更優的學名藥,病人可以向醫生或藥劑師詢問,看這種原廠藥是否已有相應的學名藥上市。病人也可以通過以下方式查找:
● 通過Drugs@FDA
● 登入FDA藥品「橙色數據庫
● 在學名藥剛通過批准後,可能不會立刻收入「橙色數據庫」。可以使用「第一通過的學名藥
責任編輯:李雯
http://www.epochtimes.com/b5/17/8/26/n9569578.htm