城中活動
2026-06-15 7:00 下午
【中醫講座】未病先防|中醫教你為家人守住健康防線
2026-06-13 3:30 下午
有人共鳴—— 一個很小、很溫柔的喪親分享聚會
2026-06-12 7:00 下午
【線上講座】家支援醫事講・老年腫瘤學新視角
疑難排解
我們有一群專業的醫護人員及相關朋友,隨時解答大家的疑難,立即提交疑問!
會員註冊
成為會員,可以第一時間接收由病患者和照顧者角度出發的資訊,立即行動!
或許你會想看
有藥冇錢醫|基層抗癌悲歌 新藥未納入安全網資助 病人或等到死
有藥冇錢醫|基層抗癌悲歌 新藥未納入安全網資助 病人或等到死 65歲的枝媽乳癌復發,其23歲的兒子Vincen […]
公院猛開藥看門口 剩藥亂丟釀海洋災難
公院猛開藥看門口 剩藥亂丟釀海洋災難 公立醫院門診輪候時間長,醫生往往開出長達半年的藥量,衍生藥餘問題。 啟德 […]
衞生事務委員會會議 (2021/06/11)
衞生事務委員會會議 (2021/06/11) 1 立法會衛生事務委員會會議,總共有八位議員就婦女健康議題發表意 […]
醫院藥劑師學會整合癌症藥物資助計劃 冀助病人選合適療程
醫院藥劑師學會整合癌症藥物資助計劃 冀助病人選合適療程 有癌症病人組織表示,不少病友都需用自費藥物,但藥費高昂 […]
我不會讓我的家人接受目前的免疫細胞治療
我不會讓我的家人接受目前的細胞療法 發文時間: 2020/08/24 文 / 黃達夫. 台北 近日報載,衛福 […]
癌症偽資訊令肖恩走上不歸路而喪命
癌症偽資訊令肖恩走上不歸路而喪命 一些不準確的癌症謠言或偽資訊,會為癌症患者帶來甚麽影響﹖治療不歸路,還是變成 […]
抗疫清潔日常
抗疫清潔日常 話咁快就過咗【抗疫生活】差唔多一個月,每日清潔消毒真係唔少得,#臨床腫瘤科專科 黃麗珊醫生同我哋 […]
港大深圳醫院教自製口罩 可達外科口罩成效九成
港大深圳醫院教自製口罩 可達外科口罩成效九成 2 1 新冠肺炎肆虐,巿面口罩難求,有巿民為自保自製口罩,但成效 […]
不容忽視氣炸鍋可能引發健康危機
不容忽視氣炸鍋可能引發健康危機 [新聞稿] 標榜無油、健康煮食的氣炸鍋,大受愛煮一族的瘋狂追捧,卻往往忽略 […]
賀建奎涉基因編輯嬰兒案 被判有期徒刑3年
賀建奎涉基因編輯嬰兒案 被判有期徒刑3年 根據法庭宣判,賀建奎干犯非法行醫罪,被判處有期徒刑3年,罰款人民幣3 […]
立法會十題:治療肺癌的新藥物
立法會十題:治療肺癌的新藥物 以下是今日(十月二十三日)在立法會會議上黃碧雲議員的提問和食物及衞生局局長陳肇始 […]
莫樹錦教授主講「抗癌漫長路」
香港中文大學 (中大) 醫學院腫瘤學系系主任莫樹錦教授,10月14日於中大以「抗癌漫長路」為題, […]











 Cellectis CEO André Choulika。(圖:AFP)
Cellectis CEO André Choulika。(圖:AFP)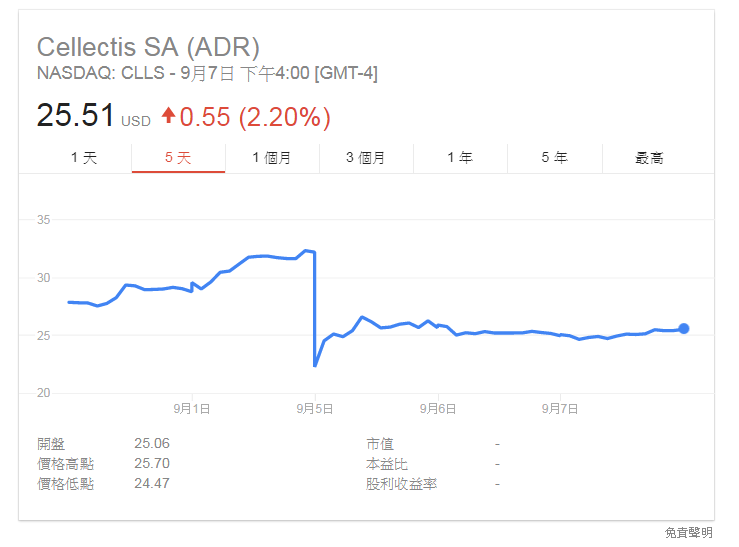 法國生物製藥公司 Cellectis 使用的 CAR-T 療法 UCART123 造成患者死亡,5 日股價暴跌 3 成,逾 4 億美元市值蒸發。(圖截自 Google)
法國生物製藥公司 Cellectis 使用的 CAR-T 療法 UCART123 造成患者死亡,5 日股價暴跌 3 成,逾 4 億美元市值蒸發。(圖截自 Google)


